Quality Management System in 2025: How eLeaP Helps You Master QMS, Nonconformance, and Change Management
Running a business without a solid Quality Management System is like flying without instruments. You might get lucky for a while. Eventually, something breaks. U.S. companies face rising pressure from regulators, customers, and supply chains. They need structured, documented, and auditable quality processes not guesswork.
That pressure explains why searches for quality management solutions have surged in recent years. Teams want software that handles deviations, tracks nonconformances, and manages change all in one place. This article breaks down exactly what a modern Quality Management System covers. It also explains how purpose-built software solves the problems that spreadsheets simply cannot.
Whether you work in life sciences, manufacturing, or financial services, the fundamentals are the same. You need visibility, accountability, and speed. Let’s dig in.
What Is a Quality Management System and Why Does It Matter?
A Quality Management System is a structured set of policies, processes, and procedures. It guides an organization toward consistent product and service quality. The ISO 9001 standard is the most recognized framework globally. However, industries like pharma and medical devices follow FDA 21 CFR Part 820 and ICH Q10 guidelines as well.
At its core, a Quality Management System answers four questions. What are our quality standards? Are we meeting them? When we fall short, how do we fix it? How do we prevent the same problem from recurring? Organizations that answer these questions consistently outperform those that don’t. Studies show companies with mature QMS frameworks see up to 20% fewer product defects.
Traditional QMS implementations relied on binders, spreadsheets, and email chains. Those approaches are slow, error-prone, and nearly impossible to audit. Modern quality management demands digital solutions. Software platforms centralize documents, automate workflows, and provide real-time reporting.
The right QMS software does more than store documents. It connects your quality processes to training, change management, and compliance tracking. That integration is what separates leading organizations from the rest.
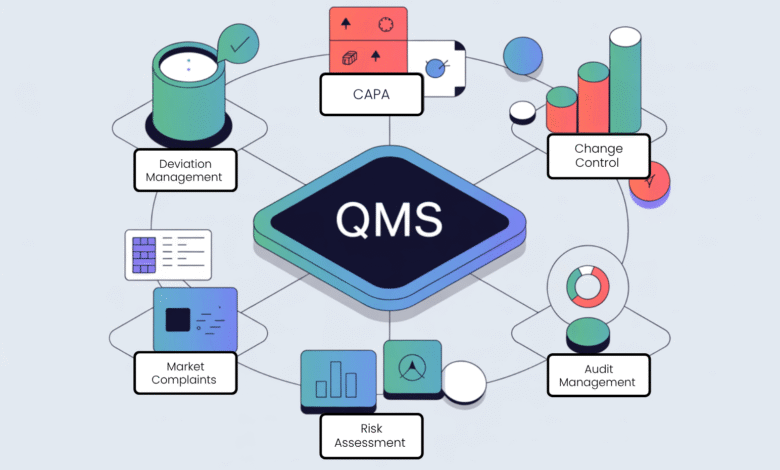
The Key Pillars of an Effective Quality Management System
An effective Quality Management System rests on several functional pillars. Each pillar supports the others. Miss one, and the entire structure weakens. Here are the core areas every QMS platform should address.
-
Document Control and SOPs
Controlled documents are the foundation of any quality program. Your standard operating procedures, work instructions, and quality policies must be current and accessible. Version control prevents employees from following outdated instructions. A QMS platform keeps every document version-tracked, time-stamped, and linked to the right approval workflow.
During an FDA audit or ISO certification review, auditors ask for document evidence instantly. A digital QMS delivers that evidence in seconds. Manual systems can take hours and sometimes days.
-
Nonconformance Management Software
Nonconformances happen in every organization. A batch fails a test. A shipment arrives damaged. A service falls below the agreed standard. How you respond defines your quality culture. Nonconformance management software gives your team a structured process for capturing, classifying, and resolving quality failures.
The right nonconformance management software does several things well. It captures the event immediately with the right data fields. It routes the issue to the correct owner automatically. It tracks resolution timelines and escalates overdue items. And it connects to your CAPA process for systematic root cause analysis.
Organizations using dedicated nonconformance management software close quality events 40% faster than those using manual processes. Speed matters because unresolved nonconformances create risk, cost, and potential regulatory exposure.
-
Deviation Management Software
In regulated industries, a deviation is any unplanned departure from an approved procedure or specification. Deviations are not the same as nonconformances though the two are closely related. Deviation management software helps teams document, assess, and resolve these departures with proper justification and oversight.
FDA inspectors and ISO auditors specifically look at how companies handle deviations. They want evidence of thorough investigation and documented decisions. They check whether similar deviations recur. Deviation management software creates an audit trail that satisfies these requirements.
Good deviation management software also connects to your risk management module. A deviation with high patient safety impact needs faster escalation than a minor process variation. Software should let you configure those risk thresholds to match your specific regulatory environment.
-
Management of Change Software
Change is inevitable. New suppliers, updated specifications, process improvements, equipment upgrades all of these trigger a change control process. Management of change software ensures that every proposed change goes through the right review and approval steps before implementation.
Without structured management of change software, organizations create risk at every change point. A process change that bypasses quality review can introduce defects. An equipment change without proper validation can violate FDA regulations. A supplier switch without risk assessment can disrupt production.
Modern management of change software automates the routing and approval workflows. It links changes to related documents, training records, and risk assessments. After implementation, it tracks effectiveness checks to confirm the change delivered the intended results.
This end-to-end visibility is critical for organizations pursuing ISO 9001, ISO 13485, or FDA compliance. Auditors expect a clear, documented change history software makes that history easy to produce.
-
LMS System Integration for Quality Training
Quality management does not end with documents and processes. Your people must understand and follow those processes. That is where an integrated LMS system becomes essential. A learning management system connected to your QMS ensures employees receive training on updated SOPs automatically.
When a procedure changes, the LMS system triggers a training assignment for every affected role. The employee completes the training. The system records the completion with a timestamp and e-signature. That training record becomes audit evidence automatically, without any manual effort.
Integrating an LMS system with your QMS also helps during onboarding. New employees complete required quality training before touching regulated processes. That integration closes a common compliance gap that standalone training platforms cannot address.
How eLeaP Brings Together QMS and LMS Under One Umbrella
Most quality platforms force organizations to stitch together separate tools for training and quality management. That creates data silos, duplicate records, and integration headaches. eLeaP takes a different approach. The platform combines a powerful Quality Management System with a built-in LMS system giving teams a single source of truth.
With eLeaP, a document change in the QMS automatically triggers a training assignment in the LMS. Employees learn about the update, confirm their understanding, and the system logs everything. There is no gap between policy change and employee awareness. That closed loop is exactly what regulators want to see.
eLeaP also offers dedicated modules for nonconformance management, deviation management, and management of change. Each module connects to the others. A deviation can generate a CAPA. A CAPA can trigger a change. A change can launch a training assignment. The entire quality lifecycle flows through one platform.
For U.S. organizations in life sciences, healthcare, or manufacturing, this integration eliminates compliance gaps. It also saves enormous administrative time. Teams spend less time chasing paper and more time improving quality.
Which Industries Need a Quality Management System Most?
Almost every regulated industry benefits from a structured Quality Management System. However, certain sectors face the highest stakes. Here is a breakdown of the industries where QMS software delivers the most value.
- Life Sciences and Pharmaceuticals: FDA 21 CFR Part 211 and ICH Q10 require robust quality systems. Deviation and nonconformance management are especially critical here. Batch record failures and out-of-specification results demand fast, documented responses.
- Medical Devices: ISO 13485 and FDA 21 CFR Part 820 govern device manufacturers. Change control and document management are under constant scrutiny during 510(k) submissions and FDA inspections.
- Manufacturing: ISO 9001 is the standard most manufacturers pursue. Management of change software becomes critical during production line updates, supplier changes, and new product introductions.
- Food and Beverage: FDA Food Safety Modernization Act (FSMA) requirements push food companies toward digital QMS solutions. Traceability and deviation tracking protect both consumers and brands.
- Financial Services: Quality management in financial services focuses on process consistency and audit readiness. Document control and training records support SOC 2 and regulatory compliance.
How to Choose the Right QMS Software for Your Organization
The QMS software market is crowded. Vendors promise compliance, automation, and ROI. Cutting through the noise requires a clear evaluation framework. Here are the factors that matter most when selecting a Quality Management System platform.
Configurability Without Coding
Your quality processes are unique. A QMS platform should adapt to your workflows not force you to adapt to its defaults. Look for drag-and-drop workflow builders, configurable forms, and role-based permissions. Avoid platforms that require professional services for every configuration change.
Validation Support for Regulated Industries
If you work in a regulated environment, your QMS software must be validated under 21 CFR Part 11 or equivalent standards. Ask vendors for their IQ/OQ/PQ documentation packages. Confirm that their e-signature features meet FDA requirements. Validation is not optional it is a compliance prerequisite.
Integration with ERP, LMS, and Other Systems
Standalone QMS platforms create data silos. The best solutions integrate with your ERP, LMS system, HR platform, and supplier portals. Native integration like the one eLeaP offers between its QMS and LMS eliminates manual data transfer and reduces the risk of compliance gaps.
Real-Time Reporting and Dashboards
Quality leaders need instant visibility into open CAPAs, overdue training, and unresolved nonconformances. Your QMS platform should deliver configurable dashboards and scheduled reports. Waiting until an audit to discover quality gaps is not a strategy it is a liability.
Implementation Support and User Adoption
The best software fails when people do not use it. Evaluate vendors on their implementation methodology, training resources, and ongoing support responsiveness. Look for dedicated customer success managers rather than generic help desk support. A smooth rollout drives adoption and maximizes your ROI.
Common QMS Implementation Mistakes and How to Avoid Them
Even well-resourced organizations make avoidable mistakes when implementing a Quality Management System. Knowing these pitfalls in advance saves time, money, and frustration.
- Boiling the ocean on day one: Organizations try to digitize every process simultaneously. This creates confusion and delays. Start with your highest-risk or most audit-visible processes. Build confidence before expanding scope.
- Skipping change management: Quality software is only as good as its adoption. Involve end users early. Communicate the ‘why’ behind the new system. Address resistance directly before go-live.
- Ignoring training integration: Deploying a QMS without connecting it to an LMS system creates a compliance gap. When procedures change, employees must know about it immediately. Manual notification processes fail under pressure.
- Underestimating data migration: Moving historical records from spreadsheets into a new platform takes longer than expected. Budget time for data cleansing, mapping, and validation before your go-live date.
- Treating validation as an afterthought: In regulated industries, system validation must happen before go-live not after. Build validation activities into your project plan from the start.
The Real ROI of a Modern Quality Management System
Quality management software is an investment. Leadership teams rightfully ask about the return. The ROI of a modern QMS extends well beyond audit readiness. Here are the measurable benefits organizations typically realize.
Reduced cost of poor quality (COPQ) is the most significant benefit. COPQ includes scrap, rework, customer returns, and warranty claims. Studies from ASQ estimate that COPQ runs between 5% and 30% of gross sales for many manufacturers. A digital QMS reduces this number by catching defects earlier and closing issues faster.
Faster audit preparation is another measurable win. Teams that previously spent weeks preparing for regulatory audits reduce that time to days with a well-implemented QMS. All evidence is centralized, searchable, and accessible with a few clicks.
Fewer repeat nonconformances reflect improved root cause analysis and corrective action. When your nonconformance management software connects to your CAPA module, root causes get addressed not just symptoms. Recurring problems decline, and quality metrics improve quarter over quarter.
Employee productivity also improves when training and quality processes live in one platform. Staff spend less time searching for documents, chasing approvals, and reconciling records. They spend more time doing valuable work. That efficiency compounds over time across the entire organization.
The Future of Quality Management: What’s Coming in 2025 and Beyond
Quality management is evolving fast. Several trends are reshaping how organizations approach their QMS strategies. Understanding these trends helps you choose a platform that will grow with your needs.
AI-assisted quality analytics is gaining traction. Platforms now use machine learning to identify patterns in nonconformance data before problems escalate. Predictive quality tools flag at-risk processes before they produce defects. This shift from reactive to proactive quality management is a significant leap forward.
Connected supplier quality management is another major trend. Organizations want real-time visibility into supplier performance, deviation rates, and corrective action status. QMS platforms are extending beyond company walls to create quality networks across entire supply chains.
Mobile-first quality workflows are becoming standard. Quality events happen on the shop floor, in warehouses, and in the field. Mobile-friendly platforms let workers capture nonconformances, complete training, and escalate deviations from any device. This immediacy dramatically improves data quality and response time.
Finally, deeper LMS system integration is a growing priority. As workforce compliance requirements increase, the line between quality management and learning management continues to blur. Platforms like eLeaP that offer native integration across both domains are positioned to lead this evolution.
Final Thoughts: Build Your Quality Foundation on the Right Platform
A well-implemented Quality Management System is not just a compliance tool. It is a competitive advantage. Organizations that manage quality proactively produce better products, serve customers more reliably, and navigate regulatory inspections with confidence.
The right QMS platform handles nonconformance management, deviation management, and management of change in one connected environment. It integrates with your LMS system to close the gap between policy changes and employee awareness. It gives leadership the real-time visibility to make data-driven quality decisions.
eLeaP delivers exactly that a unified platform that grows with your organization and supports your compliance journey from day one. Whether you are building a quality program from scratch or modernizing a legacy system, the right technology partner makes all the difference.
Ready to strengthen your quality operations? Explore how eLeaP’s integrated Quality Management System and LMS can transform the way your team manages quality, training, and compliance all in one place.